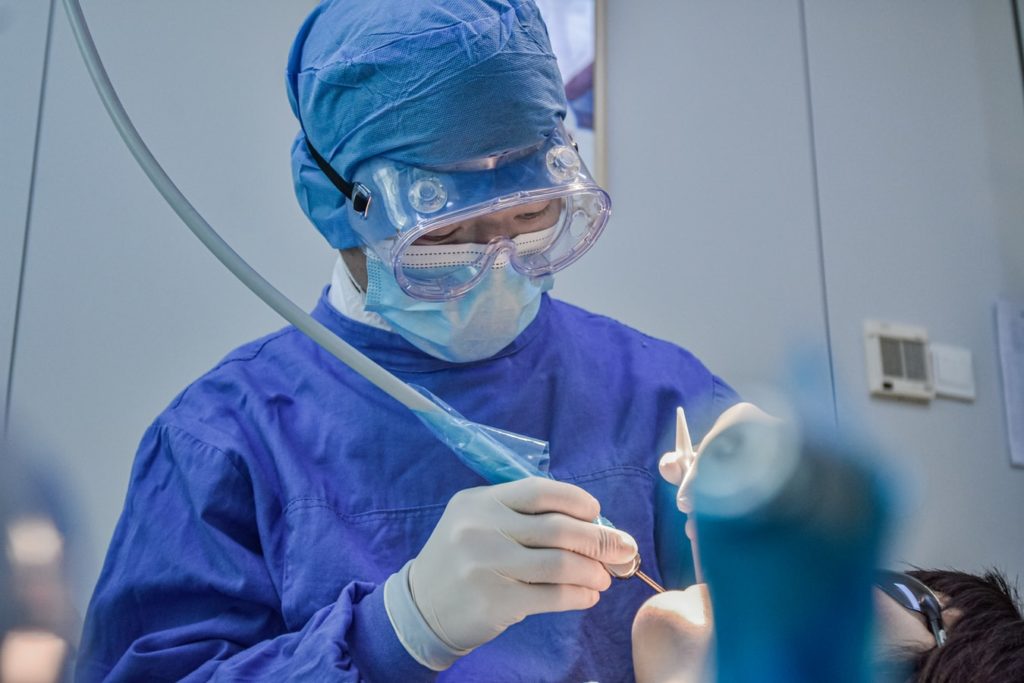
Coronavirus and dentistry
A couple of months ago, The New York Times sent out a message to dentistry professionals (Lazaro 2020). The journalists assessed the risk of infection by coronavirus in different professions and concluded that dentists and dental hygienists are the jobs with the highest chance of being infected. Policy makers and society in general were so focused on medical doctors and nurses that overlooked that people working in dental clinics are under a high risk of contagion, due to the high generation of aerosols as part of routine dental practices and the extremely short working distance between the patient and the professional. Virus particles are transmitted in small salivary aerosols that can travel a certain distance depending on their size, reaching the skin, oral cavity, nose or eyes of people around the infected person. This movement of viral particles is reduced by the patient using face masks, but this is of course not possible during dental examination and procedures.
To make things worse, a gene expression analysis in different human tissues showed that the levels of the protein ACE2 (H Xu et al. 2020), which is the receptor where coronavirus bind, is extremely high in the oral cavity, mainly in the tongue and oral mucosa. In fact, the density of the SARS-CoV-2 receptor is higher in the tongue than in the lungs. This suggests that binding to different tissues in the oral cavity may be a crucial route of viral entry in the body, and also that saliva, which bathes all oral tissues, could be an important reservoir of the pathogen (J Xu et al. 2020). This has recently been confirmed by performing diagnostic tests in saliva, showing that it is in fact a more sensitive sample than nasal swabs to detect infection (Sabino-Silva et al. 2020). This has practical implications for performing massive tests, since saliva sampling does not require a specialist to perform the collection, but it also underlines how close the dental professional is to being infected by people carrying the virus.
Furthermore, the number of infected people that appears to be completely asymptomatic has been shown to be very high and may account for almost half of the transmissions. Other studies report a much lower incidence of asymptomatic patients. It is also relevant that the highest peak in the number of viral particles appears to be produced just before the appearance of the first clinical symptoms and positive results for coronavirus are frequently documented in many patients weeks after they recovered from the disease (Rothe et al. 2020).
Although there is some controversy regarding the proportion of asymptomatic individuals and their contribution to viral transmission, many European countries decided to downsize dental practice to only provide urgent care during the pandemic. In others, treatments involving aerosol generation were minimized, although specific measures have varied. However, the situation is improving in many countries and preventive measures are already being adopted during the transition phase to normality (Ather et al. 2020).
In fact, my personal view is that dental practice and dental training may never be the same and some of the modified procedures could actually remain in the long term even after the COVID19 pandemic is over. What policies might change in the short term and perhaps be extended in the future? One of them is to consider all patients visiting the clinic as positive for this -or other- virus and ask them to wash hands upon entry in the clinic, keep social distance, minimize cash payment, etc. Given that aerosol production is difficult to be avoided, it has been proposed that we should probably get used to more strict personal protective measures such as faceshields, FFP2 masks or disposable gowns (Li & Meng 2020).
Perhaps fewer patients should be appointed, so the number of them in the waiting room is limited, and enough time is left between one appointment and the next to allow changing protective wear and ventilating well the rooms to eliminate aerosols (although this can be difficult sometimes), as well as proper disinfection of surfaces. In this regard, hydrogen peroxide and bleach are well documented to neutralize most viruses, included encapsulated coronavirus (Kampf et al. 2020). Regarding specific dental procedures, the use of rubber dam is recommended whenever possible to minimize saliva-derived contamination, as well as an efficient suction around the working area (Meng et al. 2020).
It is difficult to know which measures are really needed and whether some of them are unnecessary or will be unnecessary in the future, and current research will help establishing those. Even during the pandemic, some countries have elected to adopt extreme measures to impede transmission, and some epidemiologists have indicated that this may imply lower group immunity levels in subsequent waves of the disease; other countries have chosen to impose more flexible measures and time will indicate if this may be more successful in the long term.
Another related concern, in this case within the academic environment, is the presence of potentially infected aerosols in dental schools, given that patients are frequently treated in clinics with a fully open structure where many students are working simultaneously (Ather et al. 2020). Several dental schools are already considering creating a number of enclosed aerosol-generating side rooms within the large dental education clinics. This is an interesting initiative and could be considered in the design of new Dentistry Faculties, although it may be difficult to accommodate within the current structure of many dental teaching facilities. Other professionals consider unjustified that the design and structure of teaching facilities are conditioned by a specific infectious disease that may not be an issue in the future.
Finally, there is another measure that has been proposed by different dental associations, namely the use by the patient of an antimicrobial mouthrinse (even a nasal spray) before dental exploration and treatment, in order to reduce viral load (Caruso et al. 2020). For example, the British Endodontic Society and even the Stomatology Authorities in Wuhan have recommended 1% hydrogen peroxide as a pre-rinse mouth wash. There is also evidence that povidone-iodine is effective in neutralizing MERS-CoV (Eggers et al. 2015), another virulent coronavirus that caused an epidemic in 2012. The physico-chemical properties of Cetylpyridinium Chrloride (CPC) makes it also likely to be effective against encapsulated viruses and in fact oral rinses with this antiseptic have been shown to neutralize the influenza virus and to reduce cold symptoms in a clinical study (Popkin et al. 2017).
Chlorhexidine, having a divalent cation structure, could also provide some protection and would be interesting given its high sustantivity (it can be slowly released and remain in the mouth for hours at a working concentration), but the only study with SARS-CoV2 showed no efficacy (Kampf et al. 2020). However, only a low concentration of 0.02% was tested, and more standard 0.12 or 0.2% solutions might be more effective. For this reason, our laboratory is coordinating a clinical study in different Spanish hospitals where saliva samples are collected in COVID19 patients at baseline, and then 10 minutes, 30 minutes, 1 hour and 2 hours after a 1-minute mouthwash with either CPC, Chlorhexidine, H2O2 or povidone-iodine. A fifth group that makes an oral rinse with water will serve as control. We hope that the results will shed some light to recommend a mouth rinse protocol which could contribute to reduce the risk of infection in dentistry professionals. In Spain and Italy, people gather every day at 8 pm on their balconies and windows for a general applause to acknowledge the work and effort of sanitary personnel during the pandemic. I think we should never forget the silent and courageous work that dental practitioners are performing, and take advantage of what we have learned during this pandemic to improve the safety and working conditions of the dental community in the future.
——
Alex Mira is an oral microbiologist. He works as senior researcher at the FISABIO Institute in Valencia (Spain) and is a visiting researcher at the School of Health & Welfare, Jönköping University.
Twitter: @mira_ale
Further reading
Newspapers
1. Lazaro Gamio (2020). The Workers Who Face the Greatest Coronavirus Risk. The New York Times, March 15th 2020.
https://www.nytimes.com/interactive/2020/03/15/business/economy/coronavirus-worker-risk.html
Scientific journals
2. Ather A, Patel B, Ruparel NB, Diogenes A, Hargreaves KM. Coronavirus Disease 19 (COVID-19): Implications for Clinical Dental Care. J Endod. 2020 Apr 6. pii: S0099-2399(20)30159-X. doi: 10.1016/j.joen.2020.03.008.
3. Caruso AA, Del Prete A, Lazzarino AI, Capaldi R, Grumetto L. May Hydrogen Peroxide Reduce the Hospitalization Rate and Complications of SARS-CoV-2 Infection? Control Hosp Epidemiol. 2020 Apr 22;1-5. doi: 10.1017/ice.2020.170.
4. Eggers M, Koburger-Janssen T, Eickmann M, Zorn J. In vitro bactericidal and virucidal efficacy of povidone-iodine gargle/mouthwash against respiratory and oral tract pathogens. Infect Dis Ther 2018;7:249–59.
5. Kampf G, Todt D, Pfaender S, Steinmann E. Persistence of coronaviruses on inanimate surfaces and its inactivation with biocidal agents. J Hosp Infect. 2020;104:246-51.
6. Li ZY, Meng LY. The prevention and control of a new coronavirus infection in department of stomatology. Zhonghua Kou Qiang Yi Xue Za Zhi. 2020;55(0):E001.
7. Meng L, Hua F, Bian Z. Coronavirus Disease 2019 (COVID-19): Emerging and Future Challenges for Dental and Oral Medicine. J Dent Res. 2020;1-7. DOI: 10.1177/0022034520914246journals.sagepub.com/home/jd.
8. Popkin DL, Zilka S, Dimaano M, Fujioka H, Rackley C, Salata R et al. Cetylpyridinium chloride (CPC) exhibits potent, rapid activity against influenza viruses in vitro and in vivo. Pathogens and Immunity. 2017;2(2):253-69.
9. Rothe C, Schunk M, Sothmann P, Bretzel G, Froeschl G, Wallrauch C, Zimmer T, Thiel V, Janke C, Guggemos W, et al. 2020. Transmission of 2019-nCoV infection from an asymptomatic contact in Germany. N Engl J Med. 382(10):970–71.
10. Sabino-Silva R, Gomes AC, Siqueira W. Coronavirus COVID-19 impacts to dentistry and potential salivary diagnosis. Clin Oral Invest. 2020;24:1619–21.
11. Xu H, Zhong L, Deng J, Peng J, Dan H, Zeng X, Li T, Chen Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int J Oral Sci. 2020 Feb 24;12(1):8. doi: 10.1038/s41368-020-0074-x.
12. Xu J, Li Y, Gan F, Du Y, Yao Y. Salivary Glands: Potential Reservoirs for COVID-19 Asymptomatic Infection. First Published April 9, 2020 Letter. https://doi.org/10.1177/0022034520918518
Detta är en bloggtext. Det är skribenten som står för åsikterna som förs fram i texten, inte Jönköping University.